Development of a Cross-Linked Copepod Life-Cycle and Molecular-Genetic Assay for Examining Exposure and Effects of Coastal Use Insecticides: A Case Study with the Persistent Organochlorine Fipronil
SC Sea Grant funded researchFor the past 20 years, one of the most lethal and commonly used insecticides in coastal agriculture, golf course developments and tract housing termite control has been the organophosphate chlorpyrifos (trade name Dursban). Unfortunately, undesirable childrens´ health effects from chlorpyrifos have emerged in the past five years that have lead to its ban for homeowner use and to regulatory disfavor by the USEPA and applicators in general. As a replacement, the USEPA has licensed and recommended widespread use of the "modern" organochlorine: fipronil. Fipronil is one of the most persistent, lipophilic and toxic insecticides licensed for use since dieldrin, lindane and DDT. Surprisingly, fipronil was licensed with a very limited dataset of non-target toxicological effects on estuarine and marine fauna (US EPA Office of Pesticides, pers. comm.). Yet, fipronil is a strong neurotoxin that acts at the broad, phylogenetically conserved level of the GABA receptor. Environmentally, fipronil has a high affinity for sediment carbon and organismal lipid and a half-life of > 5 years (Hainzl and Casida 1996). These features place sediment-dependent meiobenthos and macrobenthos at especially high risk for negative population impacts from this fipronil exposure.
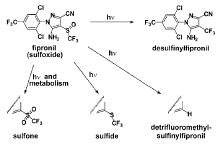
Fipronil is being used increasingly on golf courses of the south Atlantic bight as an EPA-mandated replacement for control of organophosphate-resistant (Diazinon and Dursban) mole crickets. The persistence of this compound provides satisfactory control of mole crickets with three or fewer applications per year (e.g., spring and summer). Human health concerns linked to chlorpyrifos exposure in the home have also led to increasing fipronil application as a replacement for soil-amended control of domestic termites, cockroaches, and carpenter ants (e.g., sold over the internet at http://www.doyourownpestcontrol.com/fipronil.htm or over the counter in products such as Maxforce FCTM). In South Carolina, the Charleston County municipal golf course uses fipronil for turf grass management, as do several golf courses on Hilton Head Island (National Ocean Service, unpubl.). Fipronil is relatively insoluble in water (Anon. 1999) and is readily phototransformed by sunlight to a variety of metabolites (Hainzl and Casida 1996; see Fig 1) one of which (fipronil-desulfinyl [MB 46513]) is extremely stable and is more toxic than the parent compound (USEPA 1998). Its high hydrophobicity (KOW) and long half life places it among those toxicants of highest ecological concern (hazard) in muddy estuarine settings where it is most likely to accumulate.
Figure 1: Fipronil degrades photochemically (hn) under environmental conditions to the desulfinyl derivative as the major photoproduct and the detrifluoromethylsulfinyl, sulfone, and sulfide compunds as minor products (from Hainzl and Casida 1996). Physiologically, fipronil is known to block g-aminobutyric acid (GABA) receptors and interferes with the passage of chloride ions through a GABA-gated channel. At low concentrations, it disrupts nervous system function and at higher doses causes death (Cole et al. 1993). In standard use, fipronil is a control agent for moths and grasshoppers on crops and beetle larvae in soils (Hainzl and Casida 1996). It exhibits little mammalian non-target toxicity due to its rapid degradation by the cytochrome P450 MFO system. Unfortunately, almost nothing is known of fipronil´s effects on invertebrate non-target species in estuarine receiving waters, but the emerging fipronil hazard to these systems needs to be addressed soon. The high toxicity, persistence and bio-accumulation potential of fipronil place it at the forefront of significant emerging threats to the SE coastal zone.
Research Objectives
Meiofaunal-based, reproductive life-cycle tests (Chandler and Green 2001) and modern molecular genetics techniques with meiobenthos (Schizas et al. 1997, 2001), both developed with our prior Sea Grant support, are now ready to be linked to provide a rapid and comprehensive toxicological test model for genetic and reproductive effects of persistent toxicants like fipronil. NO such genetically linked sexually reproductive models are presently available to the estuarine management community.
In this research plan, we propose to assess the lethal and sublethal effects of fipronil on the meiobenthic crustacean toxicity test model Amphiascus tenuiremis, an estuarine harpacticoid copepod that has been extensively investigated in our laboratories with other less-toxic compounds (> 16 publications in 10 years; SC Sea Grant-funded research). As a meiofaunal organism, Amphiascus tenuiremis is representative of the most abundant invertebrate taxa in the world. In specific, harpacticoid copepods are second only to nematodes in abundance in coastal sediments worldwide. They are the major meiobenthic prey of higher trophic levels (e.g., Nelson and Coull 1987, Gee 1989, Coull 1990, Coull et al. 1995), and they are the most sensitive meiobenthic taxon to pollution effects (Coull and Chandler 1992, Carman et al. 1995, Lotufo 1997). The importance of harpacticoids in the ecology of estuarine systems is unquestioned (Giere 1993).
Using a recently developed bioassay approach from the Chandler laboratory (96-well microplate full lifecycle culturing), we can track pesticide effects on genetics and reproductive development of individual copepods. Isolated microwell culture in glass facilitates a refined exposure and measurement of a pesticide? effects at the individual level on egg quality, reproductive output, developmental timing, survival from egg to sexually mature adult of offspring from single clutches, and other molecular and endocrine endpoints (e.g., ecdysone and vitellogenesis) of physiological condition. Moreover, this microplate culturing system also allows us to isolate and perform breeding experiments on the rare but evolutionarily important "toxicant resistant" individuals to determine the genetic basis of resistance and to test heritability, directly. This approach is similar to what has been done previously with Drosophila (ffrench-Constant et al. 1991, 1993); but this is the first assay of its type for an estuarine model. We will develop individual culture lines of fipronil-resistant copepods via controlled matings and genetic profiling via PCR. PCR analyses of resistant individuals will allow for straightforward development of genetic biomarkers for fipronil exposure in this and other benthic copepods. Furthermore, these techniques all make use of past progress/accomplishments from prior Sea Grant funded research.
References:
Anon. 1999. Evaluation on: Fipronil use as a public hygiene insecticide, Issue No. 187, The Health and Safety Executive, UK.
Carman KR, Fleeger JW, Means JC, Pomarico SM, McMillian DJ (1995) Experimental investigation of the effects of polynuclear aromatic hydrocarbons on an estuarine sediment food web. Mar. Environ. Res. 40: 289-318.
Chandler GT, Green AS (2001) Developmental stage-specific life-cycle bioassay for assessment of sediment-associated toxicant effects on benthic copepod production. Env. Tox. Chem. 20 (1): 171-178
Cole LM, Nicholson RA, Casida JE (1993) Action of phenyl-pyrazole insecticides at the GABA-gated chloride channel. Pestic. Biochem. Physiol. 46: 47-54.
Coull BC (1990) Are members of the meiofauna food for higher trophic levels? Trans. Amer. Microsc. Soc. 109: 233-246.
Coull BC, Chandler GT (1992) Pollution and meiofauna- field, laboratory, and mesocosm studies. Oceanog. Mar. Biol. Ann. Rev. 30: 191-271.
Coull BC, Greenwood JG, Fielder DR, Coull BA (1995) Subtropical Australian juvenile fish eat meiofauna: Experiments with winter whiting Sillago maculata and observations on other species. Mar. Ecol. Prog. Ser. 125: 11-17.
ffrench-Constant RH, Mortock DP, Shaffer CD, MacIntyre RJ, Roush RT (1991) Molecular cloning and transformation of cyclodiene resistance in Drosophila: an invertebrate GABAA receptor locus. Proc. Natl. Acad. Sci. USA 88: 7209-7213.
ffrench-Constant RH, Rocheleau TA, Steichen JC, Chalmers AE (1993) A point mutation in a Drosophila GABA receptor confers insecticide resistance. Nature 363: 449-451.
Gee JM (1989) An ecological and economic-review of meiofauna as food for fish. Zool. J. Linn. Soc. 96(3): 243-261.
Giere O (1993) Meiobenthology : the microscopic fauna in aquatic sediments. Springer-Verlag: New York. 328 pp.
Hainzl D, Casida JE (1996) Fipronil insecticide: Novell photochemical desulfinylation with retention of neurotoxicity. Proc. Natl. Acad. Sci. USA 93: 12764-12767.
Lotufo GR (1997) Toxicity of sediment-associated PAHs to an estuarine copepod: effects on survival, feeding, reproduction and behavior. Mar. Environ. Res. 44: 149-166.
Nelson AL, Coull BC (1989) Selection of meiobenthic prey by juvenile spot: An experimental study. Mar. Ecol. Prog. Ser. 53: 51-57.
Schizas NV, Street GT, Coull BC, Chandler GT, Quattro JM (1997) An efficient DNA extraction method for small metazoans. Molec. Mar. Biol. Biotech. 6(4): 383-385
Schizas NV, Chandler GT, Coull BC, Klosterhaus SL, Quattro JM (2001) Differential survival of three mitochondrial lineages of a marine benthic copepod exposed to a pesticide mixture. Env. Sci. Tech. 35: 535-538.
Staton JL, Schizas NV, Chandler GT, Coull BC, Quattro JM (2001) Ecotoxicology and population genetics: the emergence of ''phylogeographic and evolutionary ecotoxicology.'' Ecotoxicology 10(4): 217-222.
USEPA (1998) Washington DC 20460, US, Office of Prevention, Pesticides and Toxic Substances, 90 pp + 3 attachments.
FUENTE: http://www.biol.sc.edu/~coull_lab/staton/ab17.html